Phase 1 Clinical Trial Units
Early phase drug development is a complex process where many moving parts can influence the success of a program. What if there were a single partner who could proactively move your projects through the vital early stages of clinical development seamlessly?
Full-Service Phase I Clinical Units Solutions Provider
With over 30 years’ experience, Altasciences offers a simplified pathway for early clinical development so you can work with a single, integrated partner from lead candidate selection to clinical proof of concept, and beyond. We can support your entire program end to end, or you can partner with us for just one element – we offer you complete flexibility.
Our Phase I clinical trial units are flexible and adaptable for single- or multi-site clinical trials. For long or short stays, our well-appointed clinical units have all the capabilities to conduct clinical trials in a number of therapeutic areas. Expertly staffed, with dedicated USP 797 pharmacies, and with capability for specialized assessments, we are the single-source solution to your small and large molecule Phase I and II clinical trial needs.
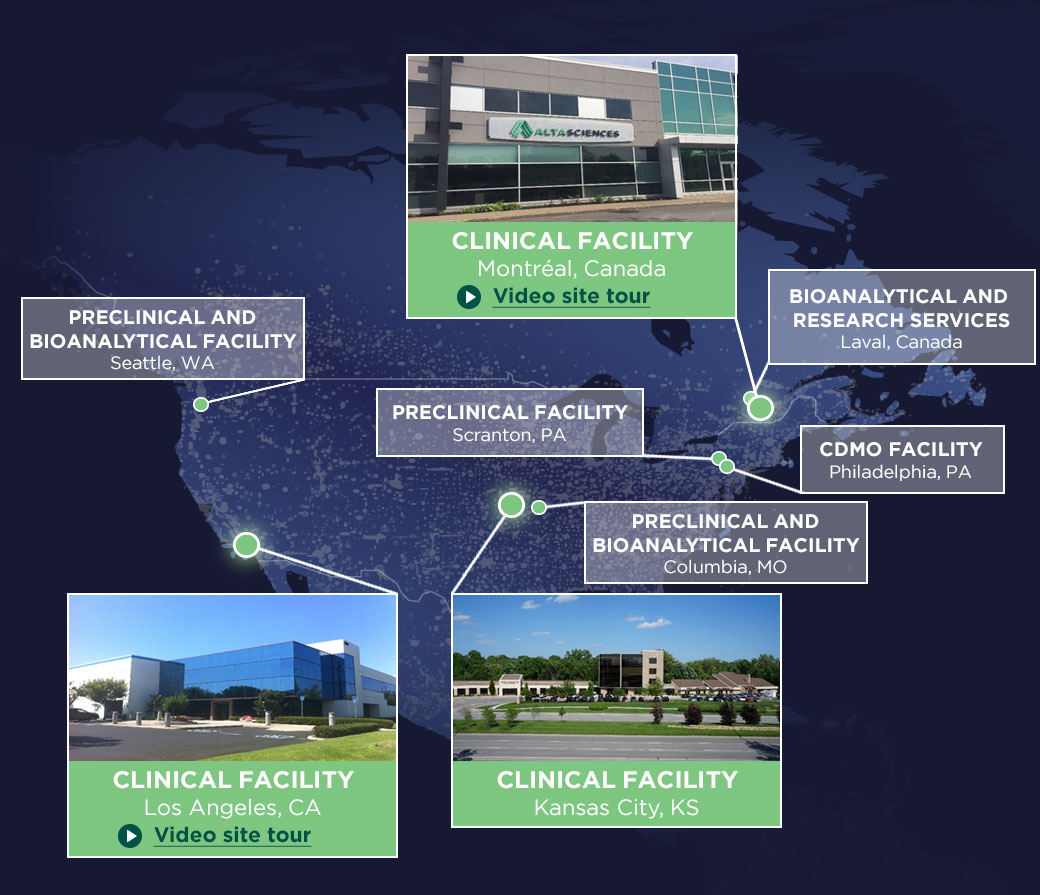
Our Early Phase Clinical Sites
- 180-bed unit with over 180 staff members
- Dedicated USP 797 pharmacy
- On-site CAP and CLIA-accredited safety reference laboratory
- PBMC processing site
- Known industry-wide for Asian ethnobridging capabilities
- Principal Investigators with anesthesiology, urology, and pediatrics specializations
- 140-bed unit with over 190 staff members
- Dedicated USP 797 pharmacy
- 36-bed specialty clinical unit for sequestering patient populations (secure access)
- Dedicated QT unit, ECGs, and Holters
- Designed to recruit and retain participants for long overnight stays
- Dedicated USP 797 pharmacy
- Neuro/CNS specialization — over 40 Human Abuse Potential (HAP) studies conducted
- Principal Investigators with psychiatry and epidemiology specializations
- 265-bed unit with over 200 staff members
- Dedicated USP 797 pharmacy
- Large database of special populations and patients for proof-of-concept (POC) arms
- Large footprint with seven individual, specialized units
- Dedicated driving simulation unit
- Neuro/CNS specialization — Driving, Human Abuse Potential (HAP), cognition, and pain
- Principal Investigators with gastrointestinal and ophthalmology specializations, and general practitioners
- Located within 5 minutes’ drive to full-service bioanalytical laboratories
Phase 1 Clinical Capabilities from Coast to Coast
Altasciences’ clinics are conveniently located close to major metropolitan areas in the U.S. and Canada, from the West Coast to the East.
Early Clinical Development Resources
With over 285 early phase clinical trials completed annually, Altasciences has developed a comprehensive portfolio of experience in early clinical development.
-
 5 Ways Altasciences Simplifies the Drug Development Process
5 Ways Altasciences Simplifies the Drug Development Process -
 8 Reasons to Choose Altasciences’ Phase 1 Clinical Trial Units
8 Reasons to Choose Altasciences’ Phase 1 Clinical Trial Units -
 Participant Retention Strategies in Phase 1 Clinical Trials
Participant Retention Strategies in Phase 1 Clinical Trials